Covid-19 mRNA vaccines - what you should know
On November 9th, 2020, Pfizer and BioNTech announced its first interim analysis (evaluable case count of 94) from the Phase 3 study of their Covid-19 vaccine candidate, BNT162b2, conducted on November 8, 2020 by an external, independent Data Monitoring Committee (DMC) from the Phase 3 clinical study. The case split between vaccinated individuals and those who receive the placebo indicates the vaccines efficacy rate of above 90%, 7 days after the second dose of the two-dose schedule required for the Pfizer-BioNTech vaccine, indicating adequate protection achieved 28 days after the initiation of the vaccination process.
Almost exactly a week later on November 16, a Boston-based pharmaceutical company, Moderna, announced its first preliminary data from the Phase 3 testing on 30,000 volunteers, with the complete data presented on Nov 30. The data showed that out of 196 cases of Covid-19 contraction, 185 of the incidences were found among those who received the placebo. Furthermore, the remaining 11 who were vaccinated did not show severe symptoms of the infection. This implied that the Moderna vaccine candidate had an efficacy rate of 94.5%.
In addition to showing efficacy rates far exceeding what experts predicted when vaccine testing began (for instance, the US Food and Drug administration considered granting emergency approval for the Covid-19 vaccine candidates if it achieved efficacy rates as low as 50%), the two front-runners for the Covid-19 vaccine (Pfizer-BioNTech and Moderna) had something else in common: both candidates were developed using the relatively novel messenger ribo-nucleic acids (mRNA) platform, different from the typical platforms that use dead or inactivated viruses or organisms to induce immunity in the recipient.

How do these vaccines work? How did these two companies develop the Covid-19 vaccines in such a short timeframe, at least compared to vaccines of the past? How is the efficacy rate (different from efficiency, by the way) so high compared to what was initially expected by many experts and the FDA themselves? What are mRNA vaccines, and how do they compare to other vaccine platforms? Why does this represent a positive step in vaccine research?
A brief overview of vaccines
The concept of vaccination is pretty simple: a vaccine’s goal is to stimulate the human immune system to produce antibodies towards a certain virus or bacteria, helping the body develop immunity to the disease without having the individual suffer from the disease itself first. In simpler words, vaccines train the body’s immune system to recognize a pathogen it has not yet encountered.
Early forms of inoculation (read: vaccination) were practiced in China and India going back to 1000 CE. That method involved grinding up smallpox scabs and blowing the matter into a person’s nostril, a process called variolation. Inoculation may also have been practiced by scratching matter from a smallpox sore into the skin. The first modern vaccine can be traced to the smallpox vaccine, created by Edward Jenner, an English physician and scientist, who used cowpox material to to inoculate patients to induce immunity to smallpox.
Many consider vaccine development as one the most (if not the most) important development in the history of preventative medication. Vaccines were certainly the 20th century’s greatest breakthrough in medicine, helping eradicate (or close to eradicating) many life-threatening infectious diseases including smallpox, polio and measles, helping increase life expectancy for the Western world from 50 to 78-85 years.
Types of Covid-19 vaccines currently in the pipeline
Apparently there are more Covid-19 vaccine candidates in various stages of clinical trials than any other infectious disease. Gavi, a public-private partnership focusing on equitable distribution of vaccines has estimated as of mid-December that more than 50 vaccine candidates are currently in clinical trials (Phase I-III) and have listed it all out here. In general there are four major categories of Covid-19 vaccine candidates, which pretty much also describe most of the vaccines currently in existence.
Whole Virus
The most classic and straightforward vaccine method is actually using the disease-causing virus itself. This is the most common method used. However, to prevent a severe illness two main approaches are used. Live attenuated viruses are basically a weakened form of the particular virus that is able to replicate itself without causing illness. Inactivated viruses are basically viruses which have had its genetic material destroyed, preventing it from replicating, but still adequate to elicit an immune response. 3 candidate vaccines from China and 1 from India are currently using the inactivated vaccine method.
Protein Subunits
Subunit vaccines involve the use of pieces of the virus - usually a particular protein or fragments - to trigger an immune response. The main benefit is that these vaccines have lower risk of causing severe illness, but at the same time could lead to a weaker immune response. To counter this, adjuvants are used to boost the immune response. The vaccine candidate from US-based Novavax is one of the few notable ones in Phase III trials using this method.
Viral Vector
These vaccines use a modified virus (the vector) to deliver genetic code for the antigen to the body’s own cells to produce them. In the case of Covid-19 the antigen is the Covid-19 spike proteins found on the surface of the virus. A common vector used is the adenovirus, which causes the common cold. Although these vaccines can induce a strong immune response compared to protein subunit vaccines, the main drawback is that patients might have existing immunity to the virus vector itself, rendering it useless or less effective in transporting the genetic instructions to the body cells. Notable vaccine candidates using this method include those from AstraZeneca/University of Oxford, Cansino and Gamaleya.
Nucleic Acid
Nucleic acid vaccines literally use genetic material itself (either RNA or DNA) to induce the body’s cells to make the antigen (spike protein for the SARS-CoV-2 virus). This method is relatively novel as no DNA or RNA vaccines have been licensed for human use (until now). The most exciting approach of all, the subject of our discussion, are the mRNA vaccines being developed by the two front-runners, the Moderna/NIAID and Pfizer/BioNTech vaccine candidates, both of which were built using the mRNA platform.
A little bit on clinical trials
Most of the vaccine candidates grabbing headlines are in Phase III clinical trials, the final of three phases that are usually required before a drug is approved by authorities (such as the U.S. Food and Drug Administration) for safe administering to the public.
Here’s a brief overview of the 3 major phases of vaccine human clinical trials:
Phase I: The vaccine candidate is usually administered to a small group of healthy people (<100) and then are monitored for the vaccine’s side-effects and to determine if the vaccine actually has therapeutic value.
Phase II: The pool of people being given the vaccine candidate is widened, and might involve people with certain characteristics (age and health) depending on the target. Usually Phase II trials last up to two years, mainly due to limitations in getting the right volunteers. The aim is to gauge the effectiveness in treating and/or preventing the illness and to determine appropriate dosing levels.
Phase III: The trial is further expanded to thousands of people to test the vaccine candidate’s effectiveness and safety across as many different subsets of the population as possible.
After the vaccines pass Phase III and get regulatory approval, monitoring is still done on the vaccines efficacy and side-effects for years after the first rollout.
How were the Covid-19 candidates developed so quickly?
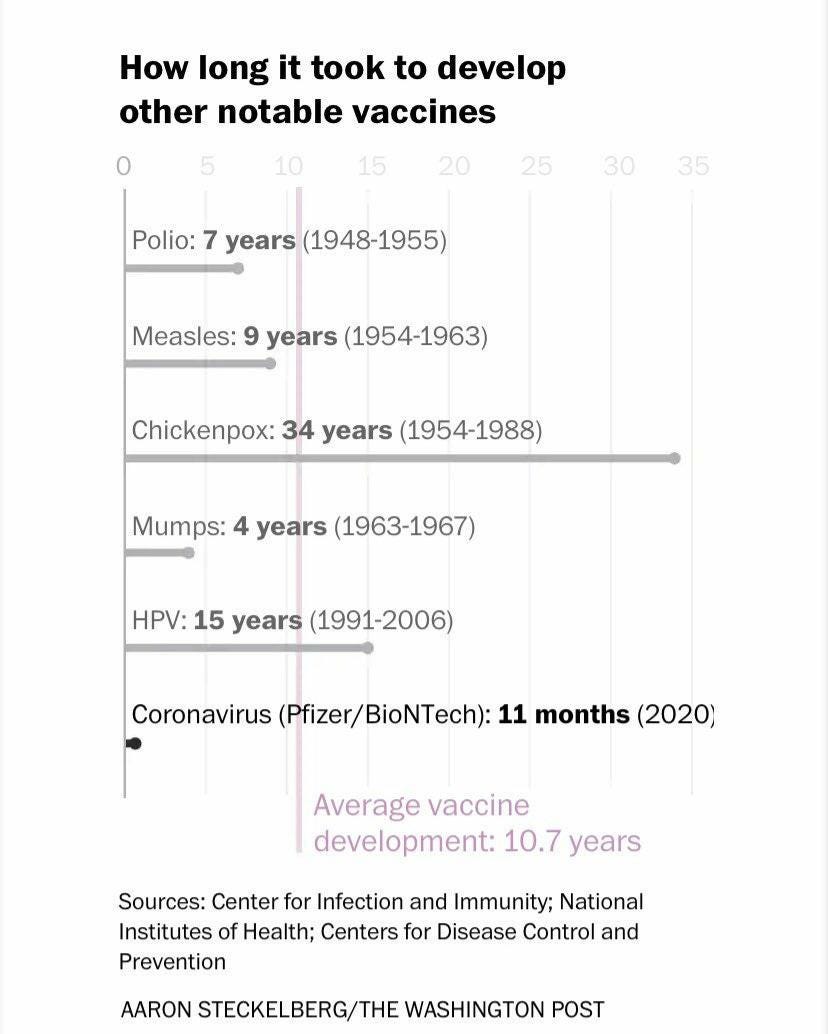
Vaccine development from the pre-clinical stage until mass production could take many years - decades in some cases. Prior to the Covid-19 vaccine, the vaccine considered to be developed the most quickly was the mumps vaccine, taking a whole four years, from viral sample collection to drug licensing in 1967. Other diseases can take much longer, with the vaccine for Chickenpox only licensed 34 years after samples were collected.
But considering the sheer economic destruction wrought upon by the Covid-19 pandemic, we might not have many years to wait for a vaccine. In January 2020, when the death toll for Covid-19 stood at a grand total of one, a team of Chinese scientists uploaded its genetic sequence to the public site, virological.org (link here). Back then, the novel coronavirus neither had a name for itself (later dubbed SARS-CoV-2), nor for the disease itself (Covid-19). Less than 10 weeks after the first SARS-CoV-2 sequence was published, Moderna and the National Institute of Allergy and Infectious Diseases had a candidate ready for a Phase I trial.
How did this happen so fast? Although not exhaustive, the following reasons are probably the biggest reasons why:
1. SARS-CoV-2 is from the coronavirus family, a family of viruses that have been well researched. We have faced coronavirus epidemics before, in the form of SARS-1 in 2002 and MERS in 2012. Many of the teams developing SARS-CoV-2 vaccines have worked on the vaccines for the SARS-1 and MERS virus. Fours strains of human coronaviruses (HCoV-OC43, HCoV-HKU1, HCoV-229E, and HCoV-NL63) continue to circulate among humans causing about 15% of common colds. These four coronavirus have a seasonal incidence, occurring during winter months, which we usually refer to as ‘flu seasons’.

2. Covid-19 is an acute disease (not a chronic one), and thus induces an immune response that can be aimed to be mimicked. Acute means that the illness develops suddenly, has distinct symptoms (think sneezing and coughing) and only lasts for a short time. After a time, the body’s natural immune response acts to eliminate the infecting virus, thus curing the body of the disease. This is what potential vaccines can aim to mimic, in contrast to some chronic diseases, like HIV, which has no natural immune response that can be mimicked.
3. The sheer scale of money pumped into the Covid-19 vaccine development. Funding has been one of the major stumbling blocks for vaccine development, mainly because of the uncertain nature of its outcome (vaccines in Phase I trials have a 22% success rate), leading to more measured less capital allocation until reliable efficacy has been established. Due to the widespread impact of the pandemic, quarters from governments, federal agencies to philanthropic organizations have all pooled together funding. Data analytics company Airfinity has put the total funding at c. £8bn (the bulk from governments) as of Dec 2020.
4. Regulatory lessons from previous crises. Lessons from the SARS-1, MERS and Ebola crisis, have caused regulators to embrace a new dexterity in approaching vaccine trials, and be more transparent in communicating with research companies on the efficacy thresholds the products need to be met. In the Covid-19 vaccine case, the US. FDA has stipulated conditional approval if the vaccines prevented infections or reduced the severity of Covid-19 in at least 50% of recipients, something it has not done before.
5. Cutting-edge approaches to vaccine development. The rapid sequencing of the SARS-CoV-2 genome enabled a lot of the vaccine-candidate frontrunners to be developed quickly. Organizations such as Johnson & Johnson, CanSino and a collaboration between AstraZeneca and Oxford University used the information from the sequencing to attach the gene for the SARS-CoV-2 spike protein to another harmless virus that ferries it to the body cells. The gene then gets expressed into the spike, allowing an immune response. Novavax is producing versions of the spike from the gene and using them directly in its vaccine candidate.
How does SARS-CoV-2 infect the body cells
Before we delve into how mRNA vaccines work, it is worthwhile to understand (to a certain extent) how SARS-CoV-2 actually infects the body. The body’s defenses system against pathogens consists of two parts: innate and adaptive. The innate system is the body’s first line of defense. It is nonspecific (acts the same way for any pathogen) and acts very quickly (almost immediately after a pathogen comes in contact with the body). Other than the skin and mucous membranes (the outer and inner linings of the body), other innate system actors include antiviral ‘interferons’ and white blood cells, which attack pathogens that manage to penetrate through the skin.
Like fellow coronaviruses SARS-CoV-1 and MERS-COV, SARS-CoV-2 affects the immune system by disrupting the actions of immune cells and interferon molecules (communication molecules between cells to trigger the protective defenses of the immune system). This explains why Covid-19 has a long incubation period (up to 14 days) compared to influenza, which typically incubates between 1-4 days. The main cause of severity in the Covid-19 disease in infected patients is thought to be due to the hyperinflammatory response induced by the SARS-CoV-2 virus. This inflammatory response might lead to ‘acute respiratory distress syndrome’ that causes shortness of breath and potentially permanent lung injury in the case of severe cases of the Covid-19 disease [5].
The adaptive immune system on the other hand, is more specific, but slower to respond compared to the innate immune system. The other advantage is that the adaptive immune system is that it is able to “remember” germs, meaning that if it encounters it a second time, the adaptive immune system can respond at a much faster rate. Usually the second infection is usually not even noticeable, or at the very least, milder.
The adaptive immune system consists of:
T lymphocytes (commonly known as T cells)
o It has features on its surface to attach to germs, and is specific for each germ, like a lock with only a particular key will fit.
o Main jobs include the use of chemical messengers to activate other immune system cells (T helper cells), detecting viral-infected/tumorous cells and destroy them (cytotoxic T cells).
o Some T helper cells become memory T cells after the infection has been suppressed. These are the crucial cells that “remember” when germs are defeated and then ready to reactivate when there is a second infection.
B lymphocytes (commonly known as B cells)
o Activated by T helper cells. The T helper cells cause B cells to match the same germs that they do, and then activates B cells to multiply and transform into plasma cells. The plasma cells produce very large amounts of antibodies and release them into the blood.
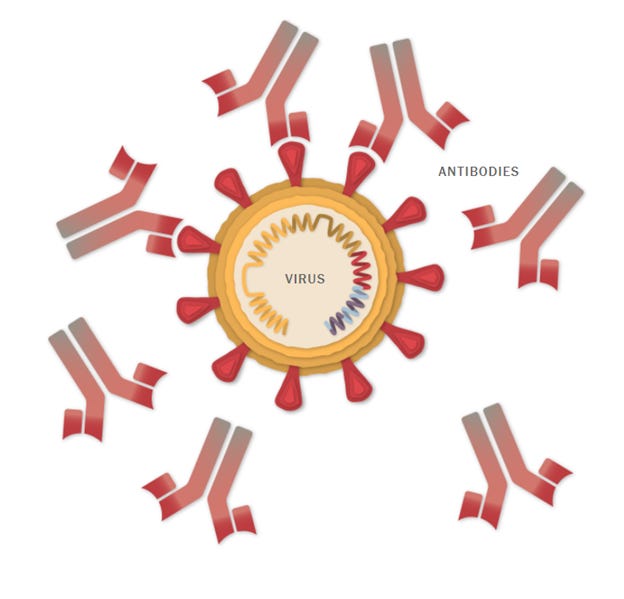
Antibodies
o Detect germs and/or its related toxins and then in bulk, attach to them. This neutralises the antigens, by basically blocking it from invading cells.
o It also allows the antigens to be more easily ingested by phagocytes (literally “eating cells”).
o Lastly it can act as tags to assist enzymes or other cells to destroy the antigens.
What is mRNA
Nucleic acids are the main information-carrying molecules in a cell. There are two general classes of nucleic acids in the body: the deoxyribonucleic acid (DNA) and the ribonucleic acid (RNA). DNAs are more chemically stable because of the lack of the highly reactive oxygen atom in its structure. RNAs are less chemically stable because it has a highly reactive oxygen atom. The other major difference is that DNA is formed when two individual strands of linked nucleotides form together to make the double helix, while RNAs are typically single stranded.
The necessary functions of life start in the genes in the DNA, which encode protein molecules (e.g. enzymes, DNA polymerases, etc), the “workhorses'' necessary to carry out various functions. When we hear of the statement “expressing a gene”, this simply means manufacturing the gene’s corresponding protein. This process is multi-layered.
The first major step in this process is transcription, where the DNA of a gene serves as a template for complementary base-pairing, to form a pre-mRNA molecule, catalyzed by an enzyme called RNA polymerase II, before forming a mature mRNA molecule.
The second major step is translation, whereby the mRNA (once out of the nucleus into the cytoplasm of a cell) is “read” within an organelle called a ribosome. The mRNA sequence is used as a template to assemble, in the particular order, the chain of amino acids that form a protein. The specific amino acid sequence, and the 3-D shape that it folds into determines the biological function of the protein. For a more in depth discussion on protein functions and folding, check out our post on DeepMind’s AlphaFold breakthrough here.
Why mRNA vaccines and how did it come about?
Conventional vaccine approaches, such as using inactivated pathogens, have been proven successful for a myriad of diseases. However, these conventional approaches do have drawbacks. For fast developing diseases like Covid-19, the main obstacle is not really the effectiveness of the vaccine, but rather the need for more rapid development and large-scale deployment. While production of conventional viral vaccines in bioreactors are a well-established process, the whole process is quite lengthy, which include steps like preparation of seed virus (usually from the live virus), fermentation, harvesting and purification which could take several months. On top of that, culturing such vaccines also require handling large volume of live viruses, which is extremely risky.
The concept of mRNA-based vaccines is quite elegant in theory - instead of synthesizing and manufacturing therapeutic proteins in bulk, why not provide information directly to the body’s cellular organelles to make the proteins themselves? The idea was basically to figure out a way to send information via mRNA directly to the patient’s ribosomes, letting the patient’s own cells manufacture the desired proteins. In the case of the Covid-19 vaccines, the aim of the mRNA vaccines is to instruct cells to produce the SARS-CoV-2 spike protein (without the whole virus itself). The protein, which will be expressed on the cell membranes or ejected into the bloodstream, will then be recognized as foreign by the body’s immune system, eliciting an immune response.
The first and most obvious advantage is that mRNA vaccines do not require injecting live vaccines into the patient’s body. mRNA is an intrinsically safe vector, as it is only a transient carrier of information to produce a specific type of protein, without directly interacting with the genome. Recall that because of the presence of the highly reactive oxygen atom in RNAs, it is inherently unstable.
The second advantage is related to manufacturing. Typical influenza vaccines can take approximately five to six months for the first supplies of approved vaccine to become available once a new strain of influenza virus with pandemic potential is identified and isolated according to the WHO. mRNA vaccines eliminate the need for a lot of the processes in manufacturing conventional vaccines such as culturing hybrid viruses or purifying attenuated viral proteins. Instead, mRNA relies on the human body to manufacture the the viral proteins themselves.
However, the mRNA therapeutics are not widespread and no mRNA vaccines have ever been approved). Why? mRNA therapeutics are not exactly new. The first recorded instance of a successful use of in vitro transcribed (IVT) (read: synthetic) mRNA in animals was published in 1990, when reporter gene mRNAs were injected into mice and protein production was detected. However, while the concept was proven and benefits widely known, early promising results did not lead to further development and widespread deployment, mainly due to:
mRNA instability: Without proper protection, mRNA degrades quite quickly inside the body, due to its inherent instability (presence of highly reactive oxygen atom), and prone to degradation by nucleases
High innate immunogenicity: foreign mRNA can trigger a strong immune response that is independent of the response to the protein it encodes. Such responses are usually hard to predict. In early tests when scientists injected synthetic mRNAs into animals, it induced such a severe inflammatory response from the immune system that the animals died almost immediately.
So how did we solve this?
Enter Katalin Kariko, a Hungarian-born biochemist at the University of Pennsylvania (UPenn), who almost lost her job, was close to deportation, and was diagnosed with cancer all at the same time, when she was deep into her research on mRNA therapeutic applications since the 1990s. Kariko even went as far as facing demotion, before she struck an unexpected partnership with fellow immunologist Drew Weissman when he moved to UPenn in 1997. Kariko and Weissman managed to identify which of the underlying nucleosides – the letters of RNA’s genetic code – was provoking the immune system, and sought to replace it with something else. The particular nucleoside was identified to be Uridine, which triggers certain immune receptors.
The major development came in 2005, when Kariko and Weissman published a study announcing a specifically modified form of mRNA, which replaced Uridine with a molecule which looked the same, but did not induced the undesired immune response (link to study here). Surely enough, when it was tested on mice, no immunogenic response was observed, and the mice lived.
An exciting development, right? Not so much apparently. At least not for the scientific community at that time.
While Kariko and Weissman published the paper, filed the patent and even established a whole company, it did not receive any traction. It however did receive attention from at least two parties, both of which would play major roles in the fight against Covid-19.
The first one was Derrick Rossi, which in the mid-2000s was a postdoctoral researcher at Stanford University, who then co-founded a biotech company called Moderna in 2010. The aim of the company was to use modified mRNA to create therapeutics, and Rossi drew insights from Kariko and Weissman’s research in their methods.
Around the same time Rossi founded Moderna, Kariko and Weissman also managed to finally license their technology to BioNTech, a German company founded by a Turkish-born medical professional by the name of Ugur Sahin. BioNTech then was mainly interested in the fields of cancer immunotherapy, cardiovascular and metabolic diseases, which are way more lucrative than vaccines, which are typically loss-leader products amongst pharmaceutical companies. That being said, both Moderna and BioNTech did venture into vaccines. Moderna began developing a potential Zika virus vaccine in 2017, while in 2018 BioNTech struck a partnership with Pfizer to develop mRNA vaccines for influenza, although funding continued to remain a key bottleneck.
Interestingly, Kariko accepted an offer in 2013 to become Senior Vice President at BioNTech after UPenn refused to reinstate her to the faculty position, even going as far as saying she “was not of faculty quality”.
The lack of spotlight and dearth of funding all changed when the Covid-19 pandemic hit in 2020. The scale of the outbreak, and then the following deluge of funding the followed allowed the mRNA vaccines to develop on an unprecedented pace.
How do mRNA vaccines work
In the case of the SARS-CoV-2 virus and other coronaviruses, the antigen of interest is the surface spike protein that the virus uses to bind and fuse with human cells. This was identified as a result of extensive research into past epidemics, including MERS and SARS-1.
So the main idea is to isolate the specific mRNA that encodes the body’s cells to produce the spike protein. Because of the instability of mRNA, the mRNA is packaged into lipid nanoparticles, allowing it to be protected from degradation by nucleases.
Upon injection into the patient’s body – usually on the arm – the packaged mRNA fuses with the body’s cells (most will be on muscle cells), enabling it to release the mRNA particles into the cells cytoplasm. The mRNA then will go through the translation process via the cells ribosomes, enabling it to manufacture the spike proteins. The spike proteins are expressed on the surface of the cell as protruding spikes, which will then be recognized by the body’s immune system as foreign.
In addition to this, when the vaccinated cells die, spike proteins and protein fragments are released, which then are engulfed by another type of immune cells called an antigen-presenting cell (APCs), which also present the fragments of spike proteins on its surface. This allows helper T cells to detect the fragments inducing other immune cells to help fight the infection.
When B cells interact with the surface of vaccinated cells, or are activated by helpter T cells, they will start to proliferate and produce antibodies to specifically target the spike protein.
The produced antibodies then serve two functions, i.e., by i) latching on to the spikes, thus neutralizing it from attaching to body cells and ii) marking the virus spikes for destruction. The APCs also can activate killer T cells to seek and destroy infected cells that display the spike protein fragment son their surfaces.
The uncertainty now lies in how long the immunity will last – how long the number of antibodies and killer T cells will remain in the body before degrading. However, the body’s immune system has specialized cells called memory B cells and memory T cells that also help retain information about the spike protein, possibly for years or even decades, allowing the body to naturally develop immunity.
To help with longer term immunity, both the Pfizer-BioNTech and Moderna approved vaccine candidates are administered in two doses. The first shot is the “prime” vaccine dose, which primes the immune system to produce antibodies to fight against the virus spike protein. The second dose, called the “booster”, will boost the immune response in the body, ensuring adequate antibody and T-cell production. The second dose is advised to be administered after 3-4 weeks of administering the first dose, as it usually takes at least 1.5-2 weeks for the body to generate enough T-cells and B-cells to create a meaningful immune response. Clinical trials show the Pfizer vaccine is most effective if the second dose is issued 21 days after the first, and the Moderna vaccine was studied with the two doses administered 28 days apart. (link)
The Pfizer/BioNTech vaccine was found to be 52% effective 10 days after the first dose, and increased to 95% 10 days after the second dose was administered, according to FDA trial data. (link).
The other sinister part of SARS-CoV-2 is in the mutations found in certain variants of the coronavirus. The mutations are found in variants that seem to transmit faster than other forms of the virus, including one variant identified in the United Kingdom and South Africa, which are causing concerns on the vaccine’s efficacy against mutated variants. So far, studies conducted have shown that the Pfizer-BioNTech vaccine has shown that its efficacy still holds against the mutated variants. The major concern here is that mutations in the RNA genome of the virus could change the appearance of certain areas of the virus, including the spike protein, which could make them less recognizable by antibodies. However, scientists do not expect the vaccines to be taken down by single mutations, mainly due to the fact that the antibodies and T cells generated target multiple parts of the virus.
Practical challenges of mRNA vaccine production
Despite it being transported in a lipid nanoparticle and having more stabilized building blocks (thanks to Kariko’s research), the two approved mRNA vaccine candidate so far need to be stored in ultra-cold temperatures to prevent them from degrading too quickly. The Pfizer-BioNTech vaccine has to be shipped in specially-designed, temperature-controlled thermal shippers that keep conditions around -70 degrees Celcius for up to 10 days. Thereafter, it needs to be stored in “ultra-low temperature freezers” for up to six months. In normal refrigeration units found in common hospitals, the Pfizer-BioNTech vaccine can be stored between 2-8 degrees Celcius for only up to five days before the vaccine degrades.
The Moderna vaccine can be shipped at -20 degrees Celcius where it can stay stable for up to six months, and can stay in normal refrigeration (2-8 degrees Celcius) for up to 30 days. The Moderna vaccine van stay stable at room temperature even for up to 12 hours, as guided on their website.
The major difference in temperatures at which the vaccines can be stored, seem to be attributed mainly to the different methods and structure of the lipid nanoparticles that encapsulate the mRNA. The specific formulations are a trade secret for these companies, so the exact reason is unknown to the public for now.
The other practical challenge is related to manufacturing capacity. With Covid-19 case numbers accelerating in most parts of the world, demand for the vaccines will definitely outstrip supply, at least for the near term, maybe even for many years to come. With only limited approvals so far for the Covid-19 vaccine candidate frontrunners, there are not many vaccines to go around. As all manufacturing activities go, hiccups are bound to happen, which can be made more severe when done at an unprecedented speed and scale, especially so near vaccine discovery. Ancillary aspects such as the supply of buffers, glass vials, pipette tips, dry ice and packaging also are crucial to ensure a smooth rollout. Adding to this challenge is the fact that vaccine manufacturers do not reveal exact production capacity, because it is a main point of negotiation when approached by buyers.
“Companies want to have flexibility in their contracts so they can balance production of various drugs and vaccines” according to Bruce Y. Lee the professor at CUNY Graduate School of Public Health & Health Policy.
mRNA therapeutics – exciting developments for the future
The success of the mRNA vaccines has led to much enthusiasm around the concept of mRNA technology for therapeutics as a whole. What makes mRNA technology very exciting is its great flexibility with respect to production and application. In theory, any protein can be encoded and expressed by mRNA. Also since the changes of the encoded protein only requires the altering of the sequence of the mRNA without affecting the physio-chemical characteristics of the molecule, diverse products can be manufactured using the same established production process. This has spurred research in mRNA’s potential to develop prophylactic and therapeutic vaccines to fight infections and cancer, as well as protein replacement therapies. For example, an mRNA sequence could help treat damaged hearts by producing a protein that stimulates blood vessel growth. Another mRNA application could have cure rare genetic diseases by encoding a missing enzyme into a patient’s body. The possibilities here seem endless.
In the realm of vaccines, the positive results from the Covid-19 mRNA vaccines bode well for other ranges of infectious diseases such as rabies, influenza and the Zika virus. However, all of these are still in the early phases of clinical trials. Moderna has currently nine prophylactic mRNA vaccines in its development pipeline, with 7 of them currently undergoing Phase I studies.
Another interesting area is in immune-oncology, which a therapeutic area that harnesses the body’s own immune system to identify and kill cancer cells in the same way the immune systems identify and targets infection from pathogens. Both BioNTech and Moderna have already developed candidate cancer vaccines that are currently in clinical trials. Both companies also have shown promising data from preliminary trails that show anti-tumour activity in people with cancer.
However, compared to vaccines, some mRNA therapeutics might need replenishment, hence possibly requiring multiple injections over time to be effective. Most of the mRNA drugs that have advanced into clinical trials so far are the ones that causes a permanent or longer-term effect (link). When repeated doses of mRNA are needed over a patient’s lifetime, side effects, such as inflammatory response to foreign RNA or buildup of lipid nanoparticles in the body could prove to be undesirable. For instance, the Covid-19 vaccine is reported to cause soreness or fever during the first few days post-injection. This is normal for most vaccines, as it is a sign that the body is mounting an immune response. However, if a drug required multiple doses over a lifetime, frequent fevers for the rest of a patient’s life might be just too much.

Some useful sources and links:
Pfizer press release on first interim analysis from Phase 3 study https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-announce-vaccine-candidate-against
Moderna data shows vaccine candidate has efficacy rate of 94.5% https://www.nytimes.com/2020/11/16/health/Covid-moderna-vaccine.html
Johnson and Johnson’s overview of the stages of the Covid-19 vaccine development https://www.jnj.com/innovation/the-5-stages-of-covid-19-vaccine-development-what-you-need-to-know-about-how-a-clinical-trial-works
How SARS-CoV-2 Infection impairs the immune system https://www.sciencedirect.com/science/article/abs/pii/S1074761320303332
Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages https://www.nature.com/articles/s41577-020-0331-4
The innate and adaptive immune system https://www.ncbi.nlm.nih.gov/books/NBK279396/
How the coronavirus affects the immune system https://www.forbes.com/sites/jvchamary/2020/11/29/coronavirus-immune-system/?sh=39e9a7221c69
Katalin Kariko’s research on suppression of RNA recognition https://www.sciencedirect.com/science/article/pii/S1074761305002116
Katalin Kariko Wiki https://en.wikipedia.org/wiki/Katalin_Karik%C3%B3#cite_note-:5-3
Story behind Moderna and Pfizer-BioNTech vaccines https://www.statnews.com/2020/11/10/the-story-of-mrna-how-a-once-dismissed-idea-became-a-leading-technology-in-the-covid-vaccine-race/
BioNTech - mRNA vaccines how they work https://biontech.de/covid-19-portal/mrna-vaccines
Paul-Ehrlich-Institut info on vaccines https://www.pei.de/EN/newsroom/dossier/coronavirus/coronavirus-node.html;jsessionid=801EB4E719E3C8C9DAF71B4618FC6F4D.intranet242
One of the first instances of nucleic acid therapeutics - injection of RNA and DNA material into a live mouse to detect expressions of proteins. https://pubmed.ncbi.nlm.nih.gov/1690918/
Pandemic influenza manufacturing timeline (from 2009 H1N1 epidemic) https://www.who.int/news/item/06-08-2009-pandemic-influenza-vaccine-manufacturing-process-and-timeline
Problems with vaccine manufacturing capacity – practical challenges https://www.statnews.com/2020/12/11/covid19-vaccine-timeline-keeps-slipping-experts-say-it-will-change-again/
mRNA technology – beyond vaccines https://www.sciencemag.org/news/2020/12/messenger-rna-gave-us-covid-19-vaccine-will-it-treat-diseases-too